Abstract
Introduction: The phase 3 MAIA study (NCT02252172) evaluated the addition of daratumumab (D) to lenalidomide and dexamethasone (Rd) in patients (pts) with transplant-ineligible newly diagnosed multiple myeloma (NDMM). At a median follow-up of 56.2 months, D-Rd prolonged progression-free survival (PFS) and overall survival (OS) versus Rd alone, despite almost half of the pts in the Rd arm who received subsequent therapy receiving a daratumumab-containing regimen as any subsequent line of therapy (Facon T, et al. Presented at: European Hematology Association 2021 Virtual Congress. Abstract LB1901). Approximately 20% to 50% of pts with MM have baseline renal impairment that can affect the choice and efficacy of therapy (Dimopoulos MA, et al. Journal of Clinical Oncology. 2016;34[13]:1544-1557). Here, we report results from MAIA for D-Rd vs Rd in pts with impaired renal function based on lenalidomide starting dose at a median follow-up of 56.2 months.
Methods: Pts with NDMM ineligible for high-dose chemotherapy and autologous stem-cell transplantation due to age ≥65 years or comorbidities were randomized 1:1 to receive D-Rd or Rd. Pts in both arms received 28-day cycles of oral Rd (R: 25 mg [10 mg recommended if creatinine clearance (CrCl) was 30-50 mL/min] on Days 1-21; d: 40 mg [20 mg if aged >75 years or body-mass index <18.5 kg/m 2] on Days 1, 8, 15, 22). Pts in the D-Rd arm also received intravenous D (16 mg/kg once weekly for Cycles 1-2, once every 2 weeks for Cycles 3-6, and once every 4 weeks thereafter). Pts in both arms were treated until disease progression or unacceptable safety events. The primary endpoint was PFS, and a secondary endpoint was OS. Renal impairment was defined as a baseline CrCl of ≤60 mL/min.
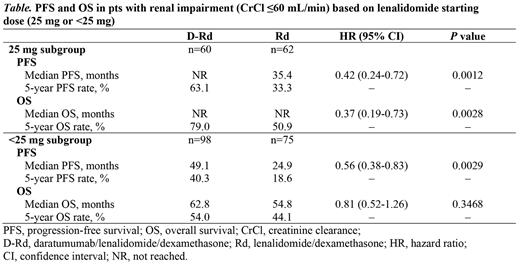
Results: 737 pts were randomized (D-Rd, n=368; Rd, n=369); 162 (44%) pts in the D-Rd arm and 142 (38%) pts in the Rd arm had renal impairment as defined. At a median follow-up of 56.2 months, in pts with renal impairment who received a lenalidomide starting dose of 25 mg (25 mg subgroup; D-Rd, n=60 [37%]; Rd, n=62 [44%]), a PFS and OS advantage was observed with D-Rd versus Rd (Table). In pts with renal impairment who received a lenalidomide starting dose of <25 mg (<25 mg subgroup; D-Rd, n=98 [60%]; Rd, n=75 [53%]), median PFS and OS were prolonged with D-Rd versus Rd (Table). Among pts in the 25 mg subgroup who died (D-Rd, n=12; Rd, n=29), disease progression was the primary cause of death in 6 (50%) pts in the D-Rd arm and 10 (34%) pts in the Rd arm. Among pts in the <25 mg subgroup who died (D-Rd, n=44; Rd, n=37), disease progression was the primary cause of death in 16 (36%) pts in the D-Rd arm and 11 (30%) pts in the Rd arm.
Conclusion: After ~5 years of follow-up, D-Rd showed a PFS improvement versus Rd in transplant-ineligible pts with NDMM and renal impairment regardless of lenalidomide starting dose. An OS advantage for D-Rd versus Rd was observed in pts with renal impairment who received a lenalidomide starting dose of 25 mg; in pts with renal impairment who received a lenalidomide starting dose of <25 mg, median OS was prolonged for D-Rd versus Rd, but the OS benefit was less pronounced. Overall, among pts with renal impairment, PFS and OS benefits of D-Rd versus Rd were observed regardless of lenalidomide starting dose; however, dose recommendations per the lenalidomide prescribing information should be followed. Many transplant-ineligible pts with NDMM may not receive a second line of therapy; our results support the frontline use of D-Rd to provide prolonged disease control in transplant-ineligible pts with NDMM and renal impairment.
Usmani: Abbvie: Consultancy; Array BioPharma: Consultancy, Research Funding; Celgene/BMS: Consultancy, Research Funding, Speakers Bureau; GSK: Consultancy, Research Funding; EdoPharma: Consultancy; Janssen: Consultancy, Research Funding, Speakers Bureau; Sanofi: Consultancy, Research Funding, Speakers Bureau; Merck: Consultancy, Research Funding; Pharmacyclics: Consultancy, Research Funding; Seattle Genetics: Consultancy, Research Funding; SkylineDX: Consultancy, Research Funding; Takeda: Consultancy, Research Funding, Speakers Bureau; Janssen Oncology: Consultancy, Research Funding; Bristol-Myers Squibb: Research Funding; Amgen: Consultancy, Research Funding, Speakers Bureau. Kumar: Novartis: Research Funding; KITE: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Abbvie: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Consultancy, Research Funding; Merck: Research Funding; Celgene: Membership on an entity's Board of Directors or advisory committees, Research Funding; Astra-Zeneca: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Beigene: Consultancy; Antengene: Consultancy, Honoraria; BMS: Consultancy, Research Funding; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Carsgen: Research Funding; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Tenebio: Research Funding; Oncopeptides: Consultancy; Bluebird Bio: Consultancy; Roche-Genentech: Consultancy, Research Funding; Adaptive: Membership on an entity's Board of Directors or advisory committees, Research Funding; Sanofi: Research Funding. Plesner: CSL Behring: Other: Advisor; Genentech: Other: Advisor, Research Funding; Oncopeptides: Other: Advisor, Research Funding; Takeda: Research Funding; Celgene: Other: Advisor, Research Funding; AbbVie: Other: Advisor, Research Funding; Janssen: Other: Advisor, Research Funding; Genmab: Research Funding. Orlowski: Amgen, Inc., BioTheryX, Inc., Bristol-Myers Squibb, Celgene, EcoR1 Capital LLC, Genzyme, GSK Biologicals, Janssen Biotech, Karyopharm Therapeutics, Inc., Neoleukin Corporation, Oncopeptides AB, Regeneron Pharmaceuticals, Inc., Sanofi-Aventis, and Takeda P: Consultancy, Honoraria; CARsgen Therapeutics, Celgene, Exelixis, Janssen Biotech, Sanofi-Aventis, Takeda Pharmaceuticals North America, Inc.: Other: Clinical research funding; Asylia Therapeutics, Inc., BioTheryX, Inc., and Heidelberg Pharma, AG.: Other: Laboratory research funding; Asylia Therapeutics, Inc.: Current holder of individual stocks in a privately-held company, Patents & Royalties; Amgen, Inc., BioTheryX, Inc., Bristol-Myers Squibb, Celgene, Forma Therapeutics, Genzyme, GSK Biologicals, Janssen Biotech, Juno Therapeutics, Karyopharm Therapeutics, Inc., Kite Pharma, Neoleukin Corporation, Oncopeptides AB, Regeneron Pharmaceuticals, I: Membership on an entity's Board of Directors or advisory committees. Moreau: Abbvie: Honoraria; Amgen: Honoraria; Janssen: Honoraria; Sanofi: Honoraria; Celgene BMS: Honoraria; Oncopeptides: Honoraria. Bahlis: Janssen: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; GlaxoSmithKline: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria; BMS/Celgene: Consultancy, Honoraria; Abbvie: Consultancy, Honoraria; Genentech: Consultancy; Amgen: Consultancy, Honoraria; Sanofi: Consultancy, Honoraria; Karyopharm: Consultancy, Honoraria. Nahi: XNK Therapeutics AB: Consultancy. Hulin: Janssen: Honoraria; Celgene/BMS: Honoraria; Sanofi: Honoraria; Takeda: Honoraria; abbvie: Honoraria. Quach: GlaxoSmithKline: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen/Cilag: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; CSL: Consultancy, Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees; Antengene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Karyopharm: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Sanofi: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding. Goldschmidt: Takeda: Consultancy, Research Funding; Sanofi: Consultancy, Honoraria, Other: Grants and/or Provision of Investigational Medicinal Product, Research Funding; Novartis: Honoraria, Research Funding; Dietmar-Hopp-Foundation: Other: Grant; Mundipharma: Research Funding; MSD: Research Funding; Molecular Partners: Research Funding; Johns Hopkins University: Other: Grant; Janssen: Consultancy, Honoraria, Other: Grants and/or Provision of Investigational Medicinal Product, Research Funding; Celgene: Consultancy, Honoraria, Other: Grants and/or Provision of Investigational Medicinal Product, Research Funding; BMS: Consultancy, Honoraria, Other: Grants and/or Provision of Investigational Medicinal Product, Research Funding; Chugai: Honoraria, Other: Grants and/or Provision of Investigational Medicinal Product, Research Funding; GSK: Honoraria; Incyte: Research Funding; Adaptive Biotechnology: Consultancy; Amgen: Consultancy, Honoraria, Other: Grants and/or Provision of Investigational Medicinal Product, Research Funding. O'Dwyer: ONK Therapeutics: Current Employment, Current equity holder in publicly-traded company, Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Research Funding; Janssen: Consultancy. Perrot: Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Sanofi: Honoraria, Research Funding; GSK: Honoraria, Membership on an entity's Board of Directors or advisory committees; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees; BMS Celgene: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Abbvie: Honoraria. Venner: Janssen: Honoraria; Amgen: Honoraria; Takeda: Honoraria; Celgene: Research Funding; Pfizer: Honoraria; Sanofi: Honoraria; BMS: Honoraria; Amgen: Research Funding. Weisel: Novartis: Honoraria; Adaptive Biotechnologies: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Karyopharm: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Sanofi: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Oncopeptides: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy; Roche: Honoraria; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; GSK: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Honoraria. Raje: Celgene, Amgen, Bluebird Bio, Janssen, Caribou, and BMS: Other. Macro: Janssen: Honoraria, Other: Travel accomodation, Research Funding; GSK: Honoraria; Sanofi: Honoraria; Celgen/BMS: Honoraria; Takeda: Honoraria, Other: Travel accomodation, Research Funding. Leleu: Bristol-Myers Squibb: Honoraria; Carsgen Therapeutics Ltd: Honoraria; Celgene: Honoraria; Gilead Sciences: Honoraria; Janssen-Cilag: Honoraria; Karyopharm Therapeutics: Honoraria; Merck: Honoraria; Mundipharma: Honoraria; Novartis: Honoraria; Oncopeptides: Honoraria; Pierre Fabre: Honoraria; Roche: Honoraria; Sanofi: Honoraria; Amgen: Honoraria; AbbVie: Honoraria; Takeda: Honoraria, Other: Non-financial support. Pei: Janssen: Current Employment, Current equity holder in publicly-traded company. Van Rampelbergh: Janssen: Current Employment. Tromp: Janssen: Current Employment, Current equity holder in publicly-traded company. Delioukina: Janssen: Current Employment.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal